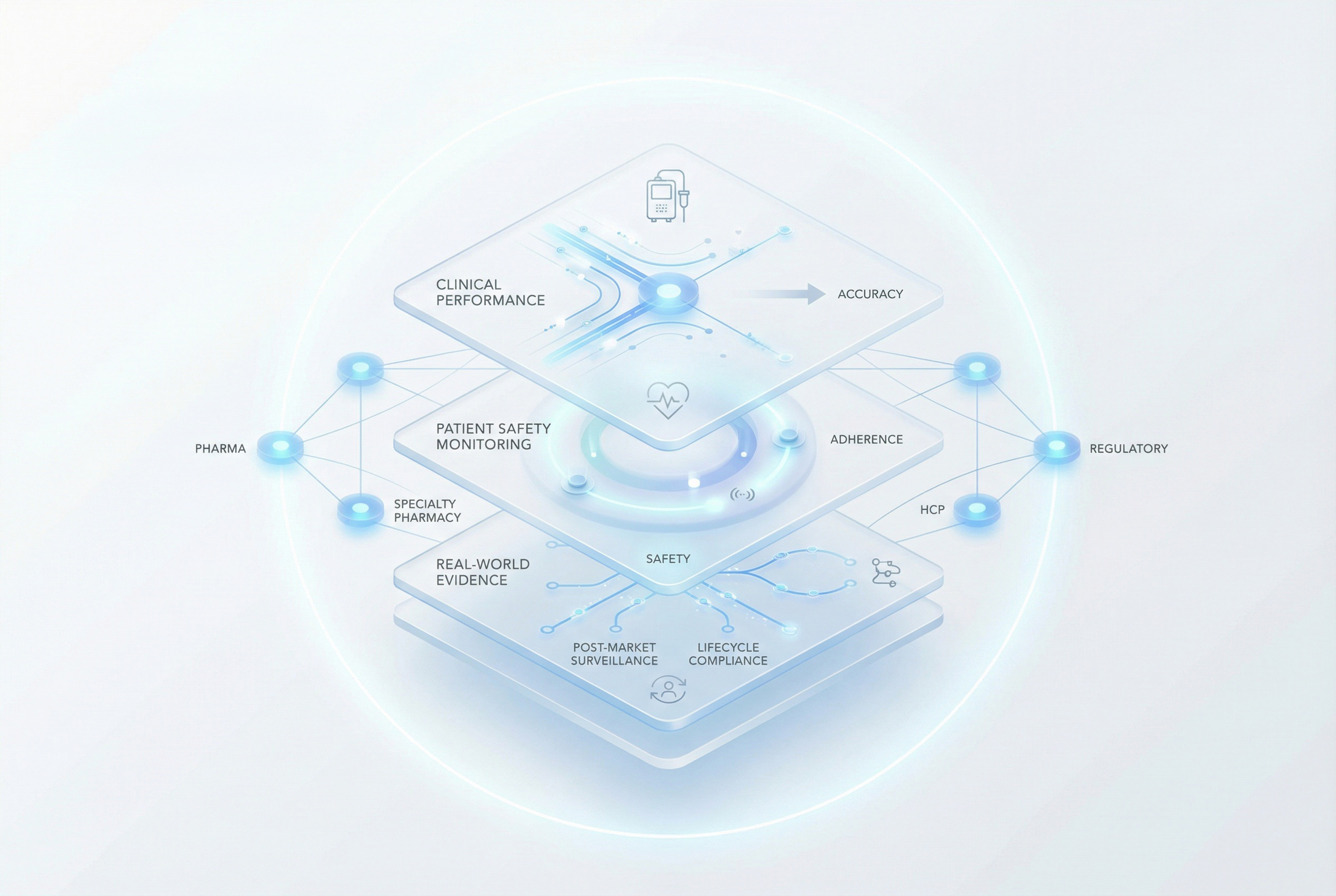
Define Scope

Successful drug delivery device development starts with identifying clinical needs, regulatory constraints, and market demands. Engaging pharmaceutical companies, specialty pharmacies, and healthcare providers ensures alignment with drug formulation, infusion parameters, and patient adherence challenges. Competitive analysis, risk assessment, and pharmacokinetic compatibility studies define critical design inputs for the infusion system.
Feasibility

Early-stage feasibility focuses on material compatibility, infusion precision, flow rate stability, and user ergonomics. Prototyping and computational modeling assess mechanical integrity, connectivity features, and usability in specialty pharmacy and homecare settings. Drug-excipient interactions, sterility requirements, and human factors engineering (HFE) inform initial concept validation.
Design

Detailed engineering involves mechanical, electrical, and firmware development to optimize performance, biocompatibility, and regulatory compliance (ISO 13485, FDA 21 CFR Part 820).
Prototyping

Rapid prototyping, functional testing, and iterative refinement ensure reliability in real-world use cases. Integration of adherence monitoring, robust connectivity across systems, and auto-calibration enhances usability for end markets including specialty pharmacies and healthcare professionals.
Validation

Preclinical testing evaluates device safety, sterility, and pharmacokinetic consistency. Simulated-use studies assess infusion accuracy, leakage, and durability. Risk management files (ISO 14971), Design History Files (DHF), and Human Factors Validation support regulatory submissions (FDA 510(k), De Novo, PMA, CE Mark).
Regulatory

Clinical validation measures infusion accuracy, patient adherence, and safety. Pharmaceutical companies and Specialty Pharmacies play a key role in trial deployment, adherence monitoring, and real-world data collection and evidence generation. Usability studies optimize patient training protocols and device ergonomics, and post-market surveillance strategies ensure continuous performance monitoring and regulatory compliance.
Manufacturing

Scaling production requires robust process validation (IQ, OQ, PQ), supply chain optimization, and quality control measures. Commercial deployment integrates with distribution networks and other commercial partnerships. Real-world evidence informs post-market improvements, driving continuous innovation and regulatory alignment.
Commercialization

Our full-suite commercialization services bridge the gap between medical device development and successful market entry. We partner with MedTech startups, established manufacturers, and biotech firms to deliver tailored strategies spanning regulatory alignment, reimbursement planning, and sales enablement, accelerating your path from FDA clearance to commercial traction, confidently and compliantly.